 
Step 5:- We keep the flask in an ice bath to cool the mixture for speed crystallization. It will destroy the excess acetic anhydride. Step 4:- We will now add 20 drops of cold water dropwise to warm water. Step 3:- We mix the solution and keep the flask in warm water for 15 minutes. 
Step 2:- Now, we will put 5 to 8 drops of 85% phosphoric acid with 6 mL of acetic anhydride into the flask. 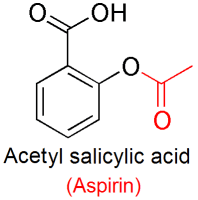
Step 1:- First, we will dry an Erlenmeyer flask and add 3 grams of salicylic acid. The synthesis of Acetylsalicylic acid is shown in the steps below: The chemical equation for this reaction is as follows: The results are Aspirin and acetic acid, where sulfuric acid or phosphoric acid acts as catalysts. It is an esterification reaction, where salicylic acid reacts with acetic anhydride, and the hydroxyl group of salicylic acid converts into an ester group. The synthesis of Acetylsalicylic acid is done by the action of salicylic acid on acetic anhydride. It is because the phenyl ring and carboxylic groups have sp2 hybridisation.īelow is the diagram that explains the structure of acetylsalicylic acid: Synthesis of Acetylsalicylic Acid When it comes to its molecular geometry, it is planar. The first component is a carboxylic acid, while the second is an ester group. Its molecule is formed by an aromatic ring with two functional groups. The IUPAC name of aspirin is 2-Acetoxybenzoic acid. Its molecular formula is \( CH_3COOC_6H_4COOH \). The empirical formula of acetylsalicylic acid is \( C_9H_8O_4 \). Acetylsalicylic Acid Formula And Structure It has various side effects, such as a high dose that may result in the ringing of ears, nausea, upset stomach, headache, etc. Its use is prohibited during pregnancy and for children with infections. And, because of this reason, the WHO’s List of Essential Medicines displays it.Īspirin is a Nonsteroidal anti-inflammatory drug, but it suppresses the normal functioning of platelets. It is one of the most effective and safest medicines and is extensively used by medications globally. When it appears in moist air, it acquires the smell of acetic acid. It is colourless to white and appears as a crystalline powder. 
It was first produced by Alsatian Chemist Charles Frederic Gerhardt in 1853 from sodium salicylate and acetyl chloride. Acetylsalicylic AcidĪlso known as aspirin or 2-Acetoxybenzoic acid, acetylsalicylic acid is a drug used to reduce pain, fever, and inflammation with a flashpoint of 482° F. So, first and foremost, let us define this acid in organic chemistry. In addition, alchemilla is also useful for muscle and joint health.Īlchemilla is the subject of more than 90 scientific publications.In this article, we will look into acetylsalicylic acid, its formula, synthesis and applications. This plant helps to maintain a good comfort before and during the menstrual cycle. The benefitsĪlchemilla contributes to digestive comfort by promoting the health of the gastrointestinal tract and the well-being of the liver. Moreover, alchemilla helps to fight against pelvic congestion syndrome by reducing the pain it generates.įinally, alchemilla is known for its anti-infectious, antibacterial, antiviral and antifungal properties. It also has vascular properties, in particular thanks to its flavonoids which improve blood circulation. Thanks to its anti-inflammatory, antioxidant, antispasmodic and sedative action, alchemilla offers numerous health benefits. In vivo studies have shown that taking alchemilla reduces endometriosis lesions and the associated inflammatory reactions. The plant contains flavonoids with antioxidant action (quercetol, hyperoside, rutoside) and salicylic acid with anti-inflammatory properties. Indissociable from the alchemists who used to collect the morning dew in the hollow of its leaves, considered as "celestial water" resulting from the condensation of effluvia circulating between the terrestrial and the celestial, the alchemilla has a mystical aspect.īelonging to the Rosaceae family, this plant is also called "women's herb" and has been reputed to cure ailments since the Middle Ages.Ĭontaining tannins (including proanthocyanidols, gallotanins and catechins) with astringent properties and a powerful haemostatic action, alchemilla stops bleeding, particularly in the event of haemorrhage or painful periods, which also enables it to indirectly prevent anaemia.Īlchemilla has many properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
